Even in a pandemic, some things do not change. This month’s Recall Roundup finds the CPSC focusing on dangers that have been front and center for some time. Specifically, the CPSC continues to focus its regulatory efforts on protecting consumers from product defects in all-terrain vehicles (ATV) and other recreational off-highway vehicles such as snowmobiles, golf carts, and utility vehicles. The CPSC recently issued a warning to consumers about the risks associated with such products, especially as more consumers look for outdoor activities during the pandemic. The warning cites to the CPSC’s Annual ATV Report of 2018, which identified almost 82,000 ATV-related injuries that required hospital treatment. Nearly one-fourth of these injuries were sustained by children under 16 years old, the highest fraction of any age group. There were also 264 ATV-related deaths in 2018, though this number is expected to rise as reporting is ongoing. So far, the CPSC has issued 15 recalls for recreational off-highway vehicles in 2020. In 2019, that figure was 20 recalls.
Unlike COVID-19, the dangers of lead are not novel as two recalls this month highlight the dangers posed by the presence of lead in consumer products, especially those targeted at children. The first recall targets an insurance company’s plush stuffed animal distributed as a promotional item. The recall covers over 635,000 units distributed over 15 years. The second recall targets a major toymaker’s water guns and covers 53,000 units distributed over five months this year. Manufacturers, distributors, and retailers should be mindful of lead levels in consumer products. Lead poisoning is a serious disease that can cause learning disabilities and long-term health problems, especially for young children.
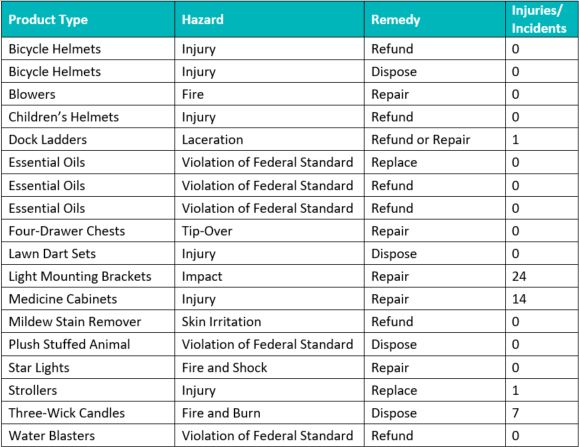
Total Recalls: 18
Hazards: Injury (6); Violation of Federal Standard (5); Fire/Burn/Shock (3); Tip-Over (1); Laceration (1); Skin Irritation (1); Impact (1)
- Partner
Kelly practices as a commercial and regulatory litigator on products liability and post M&A disputes and issues and serves as one of the firm’s Deputy General Counsel focusing on law firm ethics, conflicts, and risk management ...
Search
Recent Posts
Categories
- Advertising & Marketing
- Bankruptcy
- Class Action
- Competition/Antitrust
- Consumer Protection
- Corporate Governance
- Environmental
- General
- Health Care
- Insurance
- IP
- Labor and Employment
- Mergers & Acquisitions
- News & Events
- Patent Infringement
- Patents
- Privacy & Cybersecurity
- Product Liability
- Real Estate
- Regulatory
- Technology & E-Commerce
Tags
- 29 C.F.R. § 785.48
- 396-r
- 3D Printer
- 3D Printing
- A. Todd Brown
- A.S. Research (ASR)
- Aaron P. Simpson
- Accountability
- Administrative Agencies
- Administrative Exemption
- Advertisers
- Advertising
- Advertising Claims
- Advertising Guidelines
- Advertising Idea
- Agency Guidance
- Agency Principles
- AI Interviewing Platforms
- AI Technology Reviews
- AIA
- Air
- Algorithmic Accountability Act
- Align
- Americans with Disabilities Act
- Americans with Disabilities Act (ADA)
- Andrea DeField
- Ann Marie Buerkle
- Annual Reports
- anti-aging
- Anti-Competitive Marketplace
- Anti-Discrimination
- APEX Agreement
- Arbitration
- Arbitration Agreements
- Arizona
- Arkansas
- Arthritis
- Artificial Intelligence (AI)
- Asbestos
- Assembly Bill 51 (AB 51)
- ATDS
- Australia
- Auto-renewals
- Automatic Telephone Dialing System (ATDS)
- Automobile
- Automotive Body Parts Association (ABPA)
- Back to Work Emergency Ordinance
- biased endorsements
- Biden Administration
- Biometric Data
- Biometric Information
- Biometric Information Privacy Act (BIPA)
- BIPA
- Bitcoin
- Blockchain
- Board Diversity Disclosure
- Boards of Directors
- Bonuses
- Braille
- Branding
- Breach
- Breach of Contract
- Business Interruption
- Business Interruption Loss
- Businessowner’s Insurance
- Buy Now, Pay Later
- California
- California Air Resources Board
- California Assembly Bill 2011
- California Employment Laws
- California Fair Employment and Housing Act
- California False Claims Act
- California Labor Code
- California Legislation
- California Senate Bill 6
- California’s Unfair Competition Law
- CalRecycle
- CAMS
- Canada
- Cannabis
- CARB
- CBD
- CBP
- CCPA
- Celebrity Endorsers
- Center for Disease Control (CDC)
- CFIUS
- CGL
- Chatbot
- Children’s Advertising
- Children’s Advertising Review Unit
- Children’s Online Privacy Protection Act (COPPA)
- China
- Christopher J. Dufek
- Christopher W. Hasbrouck
- Christy Kiely
- CIPA
- Class Action
- Class Action Litigation
- Class Actions
- Clawback
- Click-to-Cancel
- Climate Change
- Climate Disclosure
- clinical trials
- Collective Action
- Colorado
- Commerce Clause
- Commercial General Liability
- Commercial Leasing
- Commercial Messaging
- Commercial Products
- Commercial Real Estate
- Commodity Futures Trading Commission
- Compensation
- Compliance
- Confidentiality
- Congress
- Connecticut
- Consent
- Consent Order
- Consumer Advertising
- Consumer Data
- Consumer Financial Protection Bureau
- Consumer Fraud
- consumer loyalty program
- Consumer Privacy
- Consumer Product Safety Act
- Consumer Products
- Consumer Products Safety Commission (CPSC)
- Consumer Protection
- Consumer Review Fairness Act of 2016 (CRFA)
- Consumer Reviews
- Consumer Rights
- Contamination
- Contract Law
- Controlled Substance Act
- Cookies
- Cookware
- COPPA
- Copyright
- Coronavirus/COVID-19
- Corp Fin
- Corporate Governance
- Corporate Reporting
- Corporate Sustainability
- Corporate Transparency Act (CTA)
- Costco
- Counterfeit Goods
- Counterfeit Goods Seizure Act of 2019
- Court of International Trade
- CPPA
- CPRA
- CPSA
- CPSC
- Crack House Statute
- CRFA
- Crypto
- Cryptocurrency
- CSPA
- Cuba
- Currency
- Customs and Border Protection
- Cyber
- Cyber Coverage
- D&O
- D&O policies
- D. Andrew Quigley
- Damages
- Data Breach
- Davidson
- Deceptive Advertising
- DEI
- Delaware
- Delivery Drivers
- DEP
- Department of Agriculture
- Department of Justice
- Department of Labor
- Development Impact Fee
- Dietary Guidelines
- Digital Assets
- digital currency
- Disclosures
- Discrimination
- Distribution
- Division of Corporation Finance
- Dodd-Frank
- DOJ
- DOL
- Duty to Defend
- Duty to Indemnify
- e-liquid products
- Eddie Bauer
- EEOC
- Electric Vehicles
- Eleventh Circuit
- Emily Burkhardt Vicente
- Employee Rights
- Employment-Based Immigration
- Endorsement
- Endorsement Guides
- Endorsement Notice
- Endorsements
- endorser monitoring requirements
- Enforcement
- Environmental Impact
- Environmental Protection Agency
- Environmental Protection Agency (EPA)
- EPA
- Epidemic
- ESG
- ESG Disclosure
- EU Regulation
- European Union
- European Unitary Patent
- EV Charging
- Exceptions
- Exclusions
- Executive Compensation Disclosure Rules
- Executive Order (EO)
- Executive Orders
- Exercise Machines
- Extended Producer Responsibility (EPR)
- FAA
- Fair Labor Standards Act
- Fair Labor Standards Act (FLSA)
- fair use
- False Advertising
- False Advertising Claims
- False Advertising Law
- False Claims Act
- Family Leave Policies
- FAR
- Fashion Accountability Acts
- FCC
- FCRA
- FDA
- Federal Acquisition Regulations
- Federal Arbitration Act (FAA)
- Federal Communications Commission
- Federal Contractors
- Federal Contracts
- Federal District Court
- Federal Government Contractor
- Federal Government Shutdown
- Federal Trade Commission
- Federal Trade Commission (FTC)
- FFDCA
- FIFRA
- Fifth Circuit
- Final Rule
- Financial Technology
- FinCEN
- FinHub
- Fireworks
- First Amendment
- Fixing America’s Surface Transportation (FAST) Act
- Florida
- Florida House of Representatives (HB 963) and Florida Senate (SB 1670)
- Florida Legislature
- FLSA
- FLSA/Wage & Hour
- FMLA
- Food and Beverage Manufacturers
- Food and Drug Administration (FDA)
- Food Delivery
- Food Safety
- Form 10-K
- Formaldehyde Standards for Composite Wood Products Act of 2010
- fractional interests
- Franchise
- Frederic Chang
- Free Trials
- FTC
- FTC Act
- Gavin Newsom
- GDPR
- General Liability
- Geoffrey B. Fehling
- Georgia
- Gift Cards
- GoodRx
- Gramm-Leach-Bliley (GLB) Act
- Green
- Green Guides
- Greenhouse Gas
- Gun Safety
- Hart-Scott-Rodino
- Hart-Scott-Rodino (HSR)
- Hashtag
- Hawaii
- Health Care
- Health Claims
- Hedge Fund
- HIPAA
- hoverboards
- human capital
- Human Rights
- IEEPA
- Illinois
- Illinois Artificial Intelligence Video Interview Act (the Illinois Act)
- Illinois Biometric Information Privacy Act (BIPA)
- Independent Contractors
- Indiana
- Indoor Mall
- Influencer Marketing
- Infringement
- initial public offerings (IPOs)
- Injury
- Insurance
- Insurance Loss
- Insurance Provider
- Intellectual Property
- Intellectual Property Licenses in Bankruptcy Act
- Inter Partes Review
- Interest Rate
- International
- International Trade Commission
- International Trade Commission (ITC)
- Internet
- Inventorship
- investigation
- INVISALIGN
- Iowa
- IP
- IPR
- Ireland
- IT
- ITC
- iTERO
- Job Posting
- Joint Employer
- Junk Fees
- Katherine Miller
- Kurt A. Powell
- Kurt G. Larkin
- Labeling
- Labeling Requirements
- Labeling Rules
- Labor
- Labor Code Private Attorneys General Act of 2004 (PAGA)
- Labor Organizing
- Labor Unions
- Land Use
- Landlord
- Latin America
- Lautenberg Act
- Lawsuit Reform Alliance of New York (LRANY)
- Lead
- Lease
- Legislation
- Leveraged Loans
- Liability Insurance Policy
- Liberty Insurance Corporation
- Liberty Mutual Fire Insurance Company
- LIBOR Discontinuation
- liquidity
- Litigation
- Live Chat
- Lost Profits
- Lost Sales
- Louisiana
- Luxury Gyms
- M&A
- Made in the USA
- Made in USA
- MagicSleeve
- Magnuson-Moss Warranty Act
- Magnuson-Moss Warranty Act (MMWA)
- Maine
- Malcolm C. Weiss
- Manufacturing
- Marketing
- Marketing Claims
- Maryland
- Massachusetts
- Matthew T. McLellan
- Maya M. Eckstein
- MD&A
- Medtail
- Membership cancellation
- Mergers & Acquisitions
- Metaverse
- MeToo Movement
- Mexico
- Michael J. Mueller
- Michael S. Levine
- microplastics
- Microplastics Litigation
- Minimum Wage
- Minnesota
- Minnesota Pollution Control Agency (MPCA)
- Misclassification
- Mislabeling
- Mission Product Holdings
- Missouri
- Mobile
- Mobile App
- Motoclick
- Multi-Family Housing Development
- Multi-Level Marketing Program (MLM)
- NAA
- NAD
- NASA
- Nasdaq
- National Advertising Division
- National Advertising Division (NAD)
- National Advertising Review Board
- National Labor Relations Act
- National Labor Relations Board
- National Products Inc.
- National Retail Federation
- Natural Disaster
- Nebraska
- Negligence Claims
- Neil K. Gilman
- Network Outage
- Nevada
- New Jersey
- New York
- New York City Department of Consumer and Worker Protection
- New York Department of Financial Services
- NHTSA
- NIL rights
- Ninth Circuit
- NLRA
- NLRB
- no-action request
- Non-Compete
- Non-Exempt
- Non-Exempt Employees
- non-fungible token (NFT)
- North Carolina
- Nutrition Labels
- Obama Administration
- Occupational Safety and Health Administration (OSHA)
- Occurrence
- Office of Labor Standards Enforcement
- Ohio
- Oklahoma
- Online Cash Providers
- Online Retailer
- Online Reviews
- Opinion Letters
- Opioids
- Opt-Out
- Oregon
- Overboarding
- Overtime
- Overtime Exemptions
- Ownership
- Packaging
- PAGA
- Pandemic
- Patent
- Patent Infringement
- Patents
- Paul T. Moura
- Pay Equity
- Pay Ratio
- Pay Transparency
- Pay-To-Play Rankings
- Penalty
- Pennsylvania
- Penny Shortage
- Personal and Advertising Injury
- Personal Data
- Personal Information
- Personally Identifiable Information
- Pesticides
- PFAS
- Physical Loss or Damage
- Policy
- price gouging
- Primary and Umbrella Policies
- Privacy
- Privacy Guidelines
- Privacy Policy
- Privacy Protections
- Procurement
- Product Liability
- Product Packaging
- Product Safety
- Prohibition on Sale
- Property Insurance
- Property Rights
- Proposed Legislation
- Proposed Rule
- Proposition 65
- Proxy Access
- proxy materials
- Proxy Statements
- PTAB
- PTO
- Public Accommodations
- Public Companies
- Purdue Pharma
- Randall S. Parks
- Ransomware
- Real Estate
- Recall
- Recalls
- Recording
- Regulation
- Regulation S-K
- Restaurants
- Restrictive Covenants
- Retail
- Retail Developers
- Retail Development
- Retail Industry Leaders Association
- Retail Litigation Center
- Retail Year in Review
- Retaliation
- Rounding
- Rulemaking
- Ryan A. Glasgow
- Sales Tax
- Salesforce
- SD8 coins
- SEC
- SEC Disclosure
- Second Circuit
- Section 337
- Section 365
- Secure and Fair Enforcement Banking Act of 2019 (“SAFE Banking Act”)
- Securities
- Securities and Exchange Commission
- Securities and Exchange Commission (SEC)
- Securities Exchange Commission
- security checks
- Senate
- Senate Data Handling Report
- Sergio F. Oehninger
- Service Contract Act (SCA)
- Service Interruption
- Service Provider
- SHARE
- Shareholder
- Shareholder Proposals
- Sign-In Wrap Agreement
- Slogan
- Smart Contracts
- SNAP
- Social Media
- Social Media Influencers
- Software
- South Carolina
- South Dakota
- Special Purpose Acquisition Companies (SPACs)
- Sponsors and Gifting
- Sponsorship
- State Attorneys General
- State Law
- State Legislation
- Store Closures
- Strategic Plan
- Subscription Services
- Substantiation
- Substantiation Notice
- Supplier
- Supply Chain
- Supply contracts
- Supreme Court
- Surveillance Pricing
- Sustainability
- Syed S. Ahmad
- Synovia
- Targeted Advertising
- Tariff
- Tax
- TCCWNA
- TCPA
- Technology
- Technology Innovation
- Telemarketing
- Telephone Consumer Protection Act
- Telephone Consumer Protection Act (TCPA)
- Tempnology LLC
- Tenant
- Tennessee
- Terms and Conditions
- Texas
- The Fair Credit Reporting Act (FCRA)
- Third-Party
- Thomas R. Waskom
- Title VII
- Tokenization
- Tokens
- Toxic Chemicals
- Toxic Substances Control Act
- Toxic Substances Control Act (TSCA)
- Trade Dress
- Trademark
- Trademark Infringement
- Trademark Trial and Appeal Board (TTAB)
- TransUnion
- Travel
- Trends
- Trump Administration
- TSCA
- TSCA Title VI
- U.S. Department of Justice
- U.S. Department of Labor
- U.S. Food and Drug Administration
- U.S. House of Representatives
- U.S. Patent and Trademark Office
- Ultra-Processed Foods
- Umbrella Liability
- Unfair and Deceptive Use
- Union
- Union Organizing
- United Specialty Insurance Company
- Unmanned Aircraft
- Unruh Civil Rights Act
- UPSTO
- US Chamber of Commerce
- US Customs and Border Protection (CBP)
- US Environmental Protection Agency (EPA)
- US International Trade Commission (ITC)
- US Origin Claims
- US Patent and Trademark Office
- US Patent and Trademark Office (USPTO)
- US Supreme Court
- USDA
- USPTO
- Utah
- Varidesk
- Vendor
- Vermont
- Virginia
- Volatile Organic Compound (VOC) Emissions
- W. Jeffery Edwards
- Wage and Hour
- Wage-And-Hour
- Walter J. Andrews
- Warning Labels
- Warranties
- Warranty
- Washington
- Washington DC
- WCAG
- Web Accessibility
- Website
- Website Accessibility
- Weight Loss
- Wiretap
- Wiretapping
- World Health Organization (WHO)
- Wyoming
- Year In Review
- Zoning
- Zoning Conversion
- Zoning Ordinances
- Zoning Regulations
Authors
- Gary A. Abelev
- Alexander Abramenko
- Yaniel Abreu
- Christian S. Adams
- Syed S. Ahmad
- Brandon Bell
- Fawaz A. Bham
- Michael J. “Jack” Bisceglia
- Jennifer L. Bloom
- Jeremy S. Boczko
- Brian J. Bosworth
- Shannon S. Broome
- Samuel L. Brown
- Tyler P. Brown
- Melinda Brunger
- Jimmy Bui
- M. Brett Burns
- Daniel J. Butler
- Matthew J. Calvert
- Grant H. Cokeley
- Abigail Contreras
- Eric S. Crusius
- Christopher J. Cunio
- Alexandra B. Cunningham
- Merideth Snow Daly
- Timothy G. Decker
- Katherine P. Deegan
- Andrea DeField
- John J. Delionado
- Stephen P. Demm
- Mayme Donohue
- Christopher J. Dufek
- Robert T. Dumbacher
- M. Kaylan Dunn
- Chloe Dupre
- Frederick R. Eames
- Andre Earls
- Maya M. Eckstein
- Rob Edwards
- Tara L. Elgie
- Clare Ellis
- Latosha M. Ellis
- Juan C. Enjamio
- Kelly L. Faglioni
- Ozzie A. Farres
- Geoffrey B. Fehling
- Jason Feingertz
- Hannah Flint
- Erin F. Fonté
- Kevin E. Gaunt
- Jane M. Geiger
- Andrew G. Geyer
- Armin Ghiam
- Neil K. Gilman
- Ryan A. Glasgow
- Tonya M. Gray
- Elisabeth R. Gunther
- Steven M. Haas
- Kevin Hahm
- Jason W. Harbour
- Jeffrey L. Harvey
- Christopher W. Hasbrouck
- Eileen Henderson
- Gregory G. Hesse
- Kirk A. Hornbeck
- Jamie Zysk Isani
- Nicole R. Johnson
- Roland M. Juarez
- James A. Kennedy, II
- Suzan Kern
- Jason J. Kim
- Scott H. Kimpel
- Elizabeth King
- Leslie W. Kostyshak
- P. Reiko Koyama
- Kurt G. Larkin
- Tyler S. Laughinghouse
- Gerry Leone
- Michael S. Levine
- Ashley Lewis
- Abigail M. Lyle
- Maeve Malik
- Eric R. Markus
- John Gary Maynard, III
- Aubrianna L. Mierow
- Gray Moeller
- Reilly C. Moore
- Michael D. Morfey
- Ann Marie Mortimer
- Michael J. Mueller
- J. Drei Munar
- Matthew Nigriny
- Michael A. Oakes
- Justin F. Paget
- Randall S. Parks
- J. Steven Patterson
- Katherine C. Pickens
- Gregory L. Porter
- Robert T. Quackenboss
- D. Andrew Quigley
- Michael Reed
- Jonathan D. Reichman
- Kelli Regan Rice
- Patrick L. Robson
- Amber M. Rogers
- Zachary Roop
- Adam J. Rosser
- Natalia San Juan
- Arthur E. Schmalz
- Daniel G. Shanley
- Madison W. Sherrill
- Kevin V. Small
- J.R. Smith
- Bennett Sooy
- Brian T. Stansbury
- Daniel Stefany
- Hak Stepanyan
- Javaneh S. Tarter
- Shauna R. Twohig
- Paige Van Oosten
- Jessica N. Vara
- Emily Burkhardt Vicente
- Mark R. Vowell
- Gregory R. Wall
- Thomas R. Waskom
- Malcolm C. Weiss
- Holly H. Williamson
- Samuel Wolff
- Jingyi “Alice” Yao
- Jessica G. Yeshman

